OraSure Technologies Inc. / AP Photo
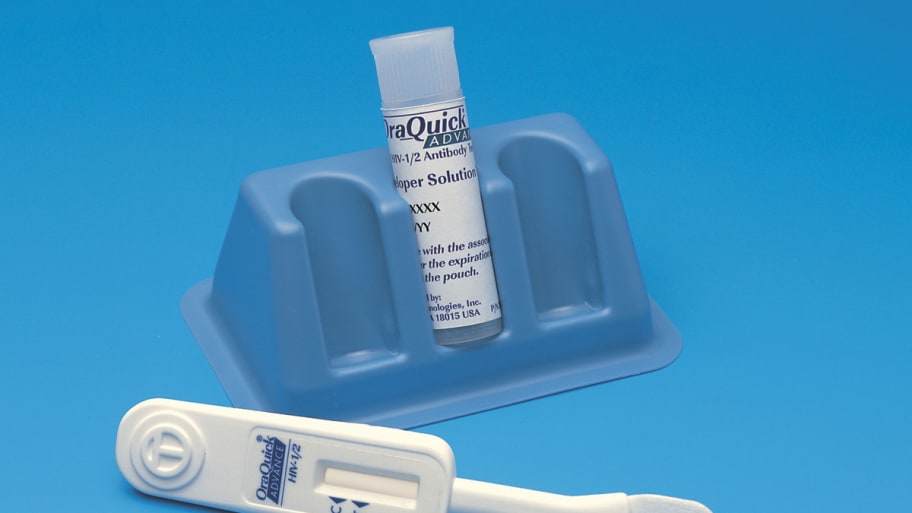
A panel of 17 experts said Tuesday that the Food and Drug Administration should give its stamp of approval to a home HIV test, the first of its kind. The test approved in a unanimous vote requires users to take a mouth swab, then gives a result within 20 minutes. “I think the information suggests that this test could be very significant in helping to stem the number of new cases of HIV that occur every year,” said Blaine Hollinger, who chairs the panel. Some critics of the test have raised questions about its accuracy, but the benefits outweigh the harm of the occasional misdiagnosis, the panel said. According to the Centers for Disease Control, more than a million Americans live with HIV, a fifth of those who are said to be unaware of his or her infection.