A new website that allows U.S. women to order abortion-inducing drugs online is under investigation by the Food and Drug Administration (FDA).
The website, Aid Access, has been quietly shipping the drugs from a pharmacy in India to women in the U.S. for the last six months, in response to what the site’s founder said was an overwhelming demand from American women.
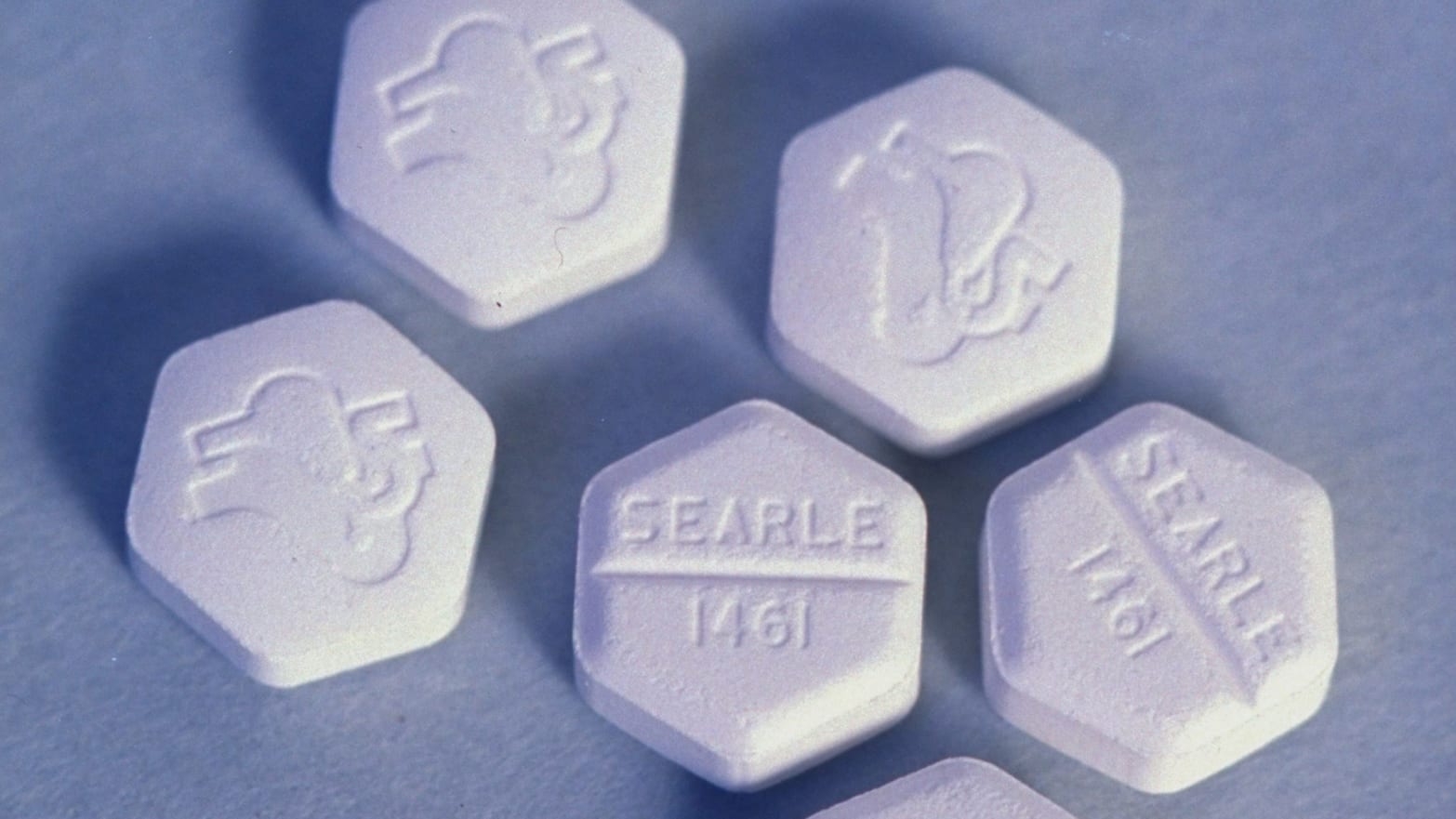
In a statement to The Daily Beast, the FDA said one of the drugs–mifepristone–is not legally available over the internet. The agency said they take claims that mifepristone is being sold online “very seriously,” and are “evaluating the allegations to assess potential violations of U.S. law.”
The website was started by the founder of a similar service, Women on Web, which ships abortion pills to women in countries outside the U.S. where abortion is illegal. The founder, Dr. Rebecca Gomperts, told The Atlantic she started Aid Access after being inundated by requests from women in America, where state laws are making in-clinic abortion increasingly difficult to access.
For women who pass the site’s online screening–they must say they are less than ten weeks pregnant, and live within one hour of a hospital, for example–Gomperts fills the prescriptions herself and sends them to a trusted pharmacy in India. She maintains that the process is legal, because the FDA allows people to import medicines for their personal use.
But the FDA currently subjects mifepristone to a strict distribution protocol, which prevents it from being sold online or in retail pharmacies. Although the drug is used in roughly a third of all first trimester abortions, and has resulted in only 22 deaths to date, it can be distributed only at pre-approved clinics, hospitals, and private practices.
Numerous medical groups, including the American College of Obstetricians and Gynecologists, have challenged the protocol, saying it is inconsistent with the requirements for other drugs with even greater risks. In a letter published in the New England Journal of Medicine in 2016, nine reproductive health experts urged the FDA to “unburden” mifepristone from the requirements.
“Medical abortion is a key component of women’s health care because it enables effective, safe, private pregnancy termination when surgical abortion is unavailable, clinically contraindicated, or personally undesirable,” the experts wrote. “ We recommend that the [protocol] be expeditiously withdrawn.”
In its statement on Sunday, however, the FDA reiterated the strict distribution restrictions, calling them “necessary to ensure the safe use of mifepristone for termination of pregnancy.”
“Mifeprex is only available to be dispensed in certain health care settings, specifically, clinics, medical offices and hospitals, by or under the supervision of a certified prescriber,” the agency said. “Mifepristone, including Mifeprex, for termination of pregnancy is not legally available over the Internet.”
Aid Access did not respond to a request for comment by the time of publication.