In 1950, Americans were closer to putting a man on the moon than they were to improving the quality of life for the disabled. One disability in particular, spina bifida, killed 90 percent of the thousands of children it affected every year.
“A child that was born with spina bifida in the 1940s and 1950s likely would not have received intervention right away,” Dr. Lisa Pruitt, medical historian and associate professor at Middle Tennessee State University, told The Daily Beast. “What doctors tended to do was wait and see if the child survived a few years [with an open lesion]; if they survived, they’d do surgery to close the defect. That was the standard of care.”
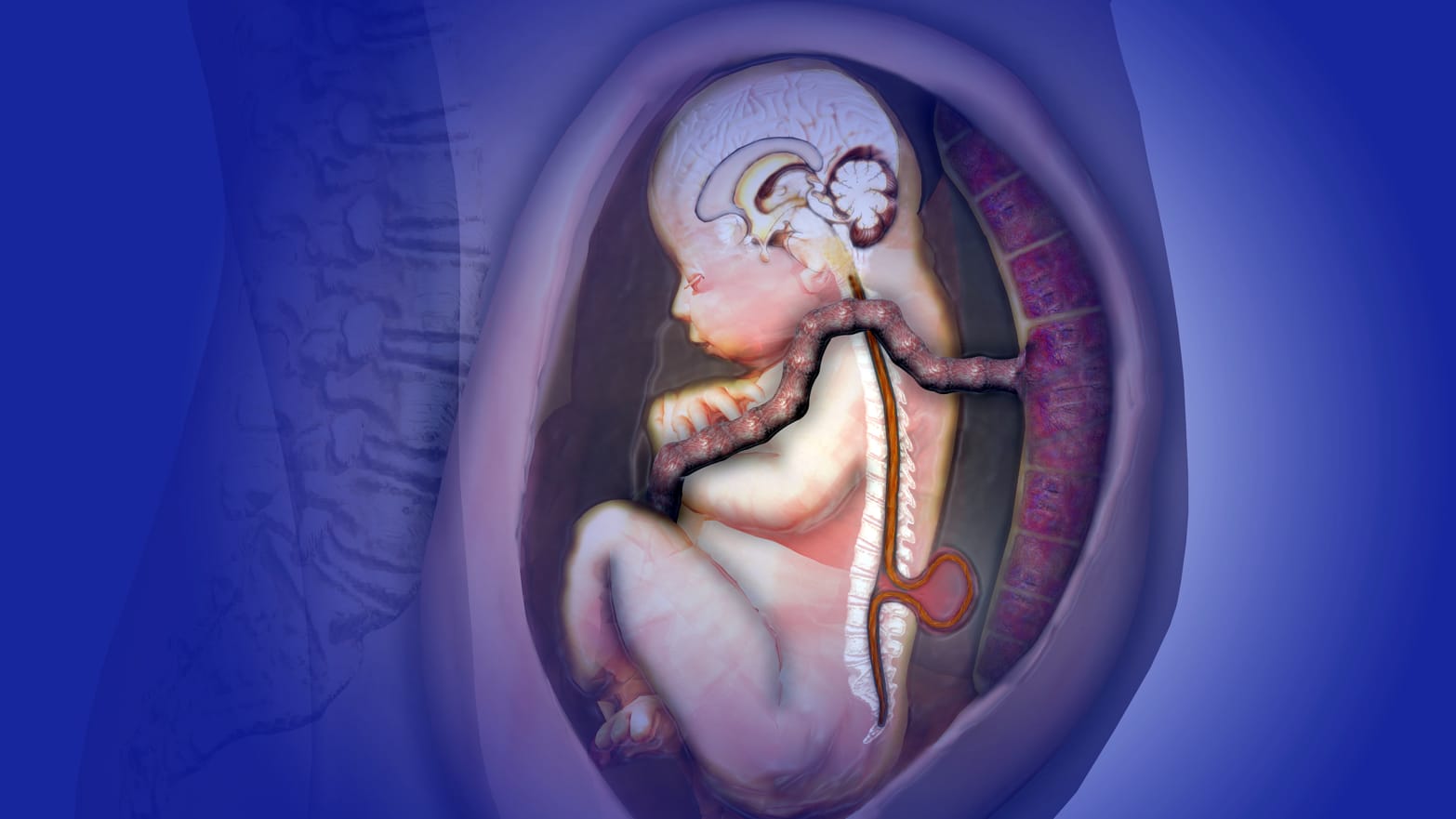
Spina bifida, a condition that still affects between 1,500 and 2,000 babies in the U.S. each year, occurs in utero before most women even know they’ve conceived. During a typical pregnancy, an embryo’s neural tube unfurls and closes by the 28th day of gestation, forming a complete brain and spinal column. But for some, the neural tube fails to close completely, resulting in a small hole in the baby’s spinal column and, later, its back (spina bifida means, quite literally, cleft spine). As the fetus grows, its spinal cord and nerves grow out of the baby’s back and are often exposed to the amniotic fluid, causing nerve damage and usually some level of paralysis from the defect area downward. A host of other medical complications, such as kidney and bladder dysfunction, brainstem malformation, and hydrocephalus usually follow.
For millennia, babies born with spina bifida typically died shortly after birth from an infection of their open spinal defect, renal failure, or hydrocephalus-related seizures. But around the mid-’60s, spina bifida slowly started becoming less fatal.
“After the polio vaccine, there started being this tremendous faith in the ability of medicine and science to fix things,” Pruitt, who was born with a less-severe form of spina bifida herself, explained. “What changed things was the beginning of the modern shunt.” Shunts, a valve placed in the patient’s head to control their hydrocephalus, started resulting in less hydrocephalus-related deaths and brain damage.
“When doctors started being able to intervene with hydrocephalus in the mid-’60s, they shifted to a philosophy of operating on babies right away, within the first few hours or days after birth,” Pruitt said. Between shunting and a new catheterization system that lessened the incidence of kidney failure, the general life expectancy of spina bifida patients grew. By the late ’70s, roughly 80 percent of babies born with spina bifida lived beyond their 5th birthday.
Today, a baby born with spina bifida is expected to reach full adulthood. Thanks to medical advancements, people born with spina bifida live active lives and generally report feeling happy and satisfied, according to information from the Centers for Disease Control and Prevention (CDC).
But the condition is still no cakewalk: Spina bifida presents lifelong challenges like paralysis and incontinence, and can be sometimes fatal for the estimated 166,000 Americans living with it today. For this reason, one collaboration of surgeons is committed to curing spina bifida completely—and they’re getting close to a breakthrough.
***
Dr. Diana Farmer grew up helping her mother teach Sunday school for disabled children. It was there she first encountered spina bifida.
“It was a really crummy disease,” Farmer, who joined the team researching stem cells at the University of California, Davis in October 2011, said. “I knew kids with spina bifida in the 1960s and nobody thought there was anything you could do for it. The main reason kids got an operation after birth was to close the [defect] and prevent meningitis. But nobody thought you’d be making them better, functionally. It just didn’t seem fair.”
Farmer recalled these children when she graduated from medical school and decided to specialize in pediatric surgery, eventually specializing in fetal surgery, which was in its infancy.
“In the early days, we didn’t want to do any [surgery] before birth unless we were sure we would have a good outcome,” she said. “Because after birth, there was no risk to the mother to operate on the baby.” Early fetal surgery was reserved for only the most fatal defects, which spina bifida no longer was.
But fetal surgery was showing incredible promise, despite the risks: In 1981, at the University of California at San Francisco (UCSF), Chief of Pediatric Surgery Michael Harrison was the first to perform a successful fetal surgery on a human, successfully treating a potentially fatal blockage in the fetus’ urinary tract. Farmer, who trained under Harrison at UCSF, was amazed. Could fetal surgery have the same life-altering effects for patients with spina bifida?
With a group of colleagues, Farmer set out to find if it could. In 2002, the Management of Myelomeningocele Study (MOMS) began.
Dr. Scott Adzick, now surgeon in chief at the Children’s Hospital of Philadelphia (CHOP) and a senior author of the study, explained: “At multiple medical centers, with the help of the NIH, we decided to do a randomized trial of myelomeningocele closure before and after birth.” Prospective patients, having undergone an intense screening process, would be randomly selected as part of the trial to undergo either a standard myelomeningocele closure after birth, or undergo fetal surgery at one of three hospitals across the nation. If the patient was selected for fetal surgery, doctors would deliver the baby by cesarean section between 22 and 26 weeks gestation, repair the defect, and then place the fetus back inside the mother to continue to grow. Both mother and child were kept under careful observation at the hospital until it was time to deliver, again by C-section, at around 36 weeks.
“Our goal was, within seven years, to get 100 patients on each ‘limb’ of the treatment centers,” Adzick said. For seven years, each hospital performed fetal surgeries back-to-back, with little idea of whether the surgeries were a success. Evaluations of the baby were done at 12 and 30 months, by pediatricians who were unaffiliated with the research team. “We were in the dark for about seven years in regard to the results,” Adzick said.
The results were beyond what anyone expected. At 30 months, less than half of the toddlers in the prenatal surgery group needed a shunt, as opposed to 82 percent of toddlers in the postnatal group. Astoundingly, children in the prenatal surgery group were twice as likely to walk without assistance. And for a quarter of babies that had received the prenatal surgery, the hindbrain herniation that doctors detected on ultrasound in utero had actually reversed or vanished completely.
“Before this, everyone thought that spina bifida was a fixed defect—you were just paralyzed for life,” Farmer said. “But I think the most important part that came out of the MOMS trial was that there’s some hope, there’s some plasticity with the spinal cord, and if we intervene early, we can make a difference.”
***
Six years later, fetal surgery for spina bifida has become the standard of care. Adzick, who would later found the Center for Fetal Diagnosis and Treatment at CHOP, has performed over 300 prenatal spina bifida repairs at his hospital alone. Hospitals in Cincinnati, Nashville, Houston, and others now offer fetal surgery for low-risk mothers and their babies.
But Farmer isn’t satisfied. Despite the incredible outcomes, Farmer points out that 60 percent of fetal surgery patients still aren’t able to walk independently. “It’s good,” she said, “but it’s not a cure.”
When the MOMS trial concluded in 2011, Farmer went back to the lab. “We started asking, what are some ways we can augment this repair before birth to make things even better?”
Farmer, along with a small team of stem cell scientists and tissue engineers, believes stem cells are the answer. For decades, researchers have used stem cells—multicellular organisms that promote cell growth—to improve conditions like bone injury and leukemia. Applied to fetuses with a major spinal defect, it may mean the difference between walking and using a wheelchair.
“We believe that for this purpose, we really only have to influence the already-rich regenerative environment that the fetus is in,” Farmer said. “The process of spinal cord development is happening in utero already—we just have to help it along.”
In 2013, Farmer’s team started operating on sheep fetuses, the same way they did before the MOMS trial started. After inducing a spinal cord injury in the sheep, the team applied placental-derived stomal cells to the fetuses in the area of injury. “We discovered that if we gave some lambs the stem cells in the matrix, they were able to walk,” Farmer said. “That got us excited.”
In August, the team applied the same treatment to a pair of English bulldog puppies, both born with spina bifida and unable to walk or wag their tails on their own. Four weeks after surgery, the two were able to run and play like typical dogs.
“It’s early,” Farmer said, “but it’s certainly enough that we’ve gotten excited about the possibility [of] curing the paralysis associated with this.”
Within the next year, Farmer hopes to test out this revolutionary treatment in human subjects. Similar to the MOMS trial, Farmer’s team would apply the stem cell treatment to the fetus between 22 and 26 weeks of gestation and then deliver just before term.
“The process of drug development is always a long one,” she said. “We want to make sure this is safe. But we’re about to file with the FDA to get us started down the path to test this in humans. We’re hoping it won’t take more than a year to get started.”
Although it seemed impossible a century ago, scientists and engineers have managed to put a man on the moon and deliver him safely back home. With time and proper funding, scientists and engineers may now be able to cure one of the oldest and most debilitating defects in human history. And with that comes the potential to cure a host of other conditions as well.
“I believe the day will come where we can treat everything before birth, from high blood pressure to cystic fibrosis,” Farmer predicted. “Between stem cell therapy, gene therapy, and surgical therapy, why treat a disease when you can prevent it?”