UPDATE: According to a statement from WHO Friday, Merck & Co.’s VSV-EBOV vaccine has proven “highly effective,” with 100 percent efficacy in clinical trials. The vaccine was developed by an Ames-Iowa based pharmaceutical called NewLink and commissioned by the Public Health Agency of Canada.
The following was originally published on Wednesday, January 7, 2015.
The need for an Ebola vaccine in West Africa has never been greater.
While public interest in Ebola continues to dwindle, the epidemic itself continues to soar. On Tuesday, the World Health Organization reported more than 20,000 cases of the disease in West Africa, nearly half of them in Sierra Leone. The death toll, which experts believe has been significantly undercut by secret burials, stands at 7,905. With a mortality rate of 70 percent, the more cases that arise, the deadlier this epidemic becomes.
Although a few smaller companies have become involved in the race for a vaccine, three major pharmaceutical makers are taking the lead—each pursuing a different vaccine. The trials are unprecedented for a variety of reasons, including the rapid timeline (trials of this nature generally take three to four years). Each individual race involves an unusual collaboration between researchers, manufacturers, and public-health entities. Together, the teams are working 24 hours a day for a product that promises much higher risk than it does profit.
Here’s what you need to know about the Ebola vaccine front-runners.
1. Johnson & Johnson (J&J)
Status: Phase 1, first in human trials at University of Oxford Type: Two-shot vaccine, specifically targeting the Zaire species of Ebola
The U.S. drug giant made headlines Tuesday with the news that human trials of its experimental Ebola vaccine are now underway in the U.K. Although a late entry to the race—its announcement came in September—J&J has quickly stolen the spotlight. In October, the company pledged more than $200 million to fast track its unique vaccine, which takes a two-pronged approach to immunizing a patient against Ebola.
Seventy-two adults between the ages of 18 and 50 are participating in the trial, led by the pediatrics department at Oxford. The accelerated vaccine utilizes two different treatments—the first to “prime” the patient’s immune system, and the other to “boost” it—each manufactured by separate international biotechnical companies (one, Denmark-based Bavarian Nordic; the other, Netherlands-based Crucell). If successful, Johnson & Johnson plans to have as many as 400,000 doses available by April, and up to 5 million by the end of the year.
2. GlaxoSmithKline (GSK)
Status: Phase 2, administering the vaccine to thousands of Ebola workers Type: Two different specific strains of Ebola delivered via a chimpanzee carrier virus
British drug behemoth GlaxoSmithKline got a head start on J&J, partnering with the National Institute of Allergy and Infectious Diseases (NIAID) to begin human trials of an experimental Ebola vaccine in late August. GSK’s involvement springs from Okairos, a Swiss-Italian biotechnology company that it acquired in 2013. The vaccine was co-developed by Okairos and a team of American scientists led by Dr. Nancy J. Sullivan, chief of Biodefense Research Section in NIAID’s Vaccine Research Center.
The NIAID/DSK vaccine is different from J&J’s in that it contains segments of the Ebola virus’s genetic material from two different strains: Zaire (this current outbreak) and Sudan. The vaccine is delivered through a “carrier virus” that causes a common cold in chimpanzees but does not affect humans. Twenty adults in Bethesda, Maryland, were injected with the vaccine for the Phase 1 trial in August. The trials produced positive results, published in The New England Journal of Medicine in November. Four weeks after the injections, all 20 of the participants had developed the antibodies needed to stave off the infection. The next phase of the trial consists of vaccinating Ebola workers on the front lines. A date has yet to be announced.
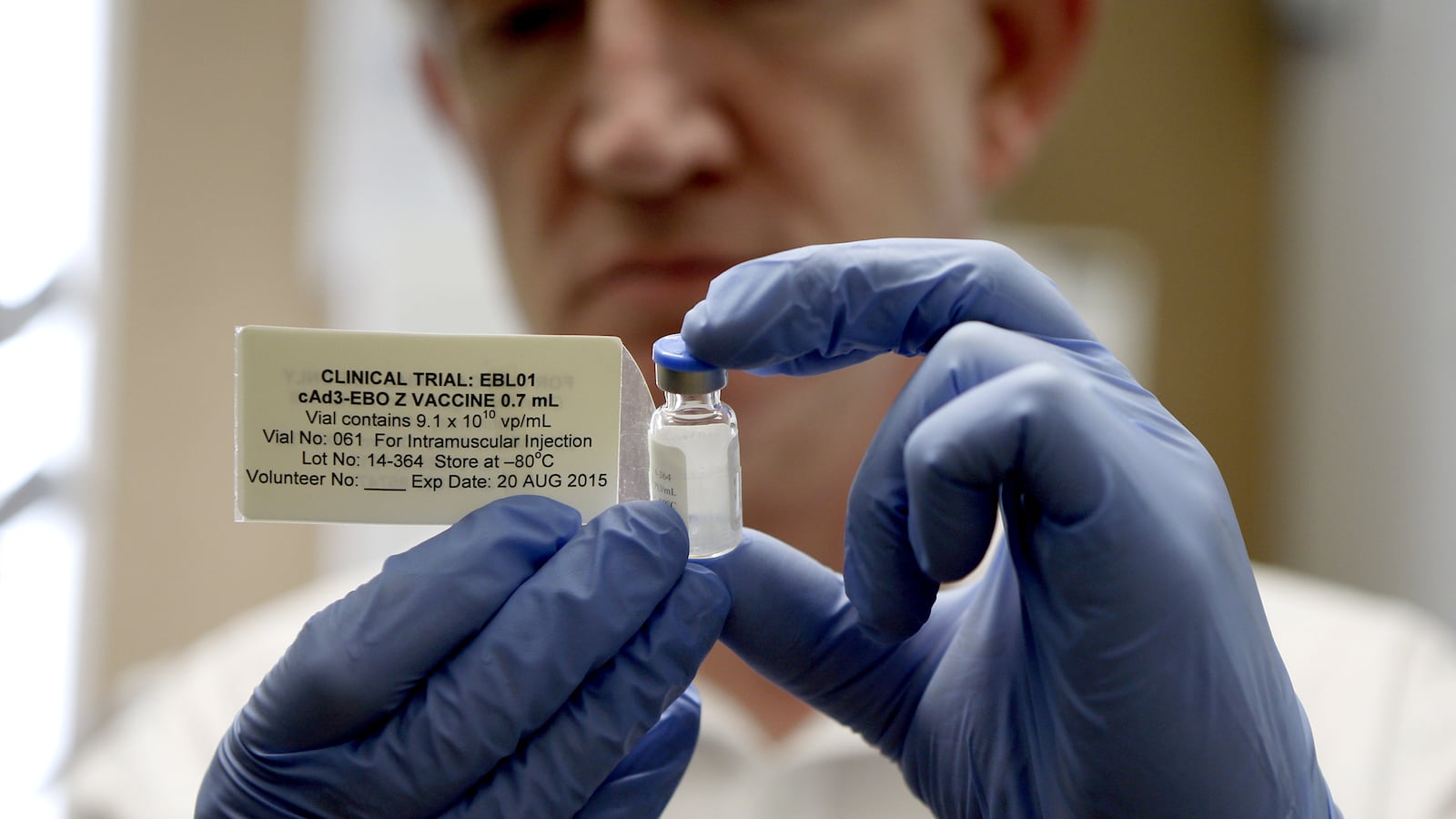
3. Merck & Co.
Status: Phase 1, restarting first in human trials at lower dose due to complaints of joint pain Type: Genetically engineered version of vesicular stomatitis virus (VSV)
Decidedly the underdog in this fight is American pharmaceutical company Merck & Co., whose effort rests on a tiny Ames, Iowa-based biotechnology company called NewLInk Genetics Corp. Merck bought the rights to NewLink’s vaccine in late November, a treatment that was initially commissioned by the Public Health Agency of Canada in 2011 for $200,000.
NewLink’s vaccine, VSV-Zebov, is “based in part on a genetically engineered version of vesicular stomatitis virus (VSV).” The gene for the outer protein of the VSV virus, which is incredibly rare in humans, is replaced with a segment of the gene from the Ebola Zaire species. The company began Phase 1 first in human trials in December, after getting approval from the Federal Drug Administration. But the trials, conducted at the University Hospitals of Geneva, were halted when volunteers reported joint pain as a result of the injection. The Phase 1 trials are set to resume this week, using a lower dose of the vaccine.