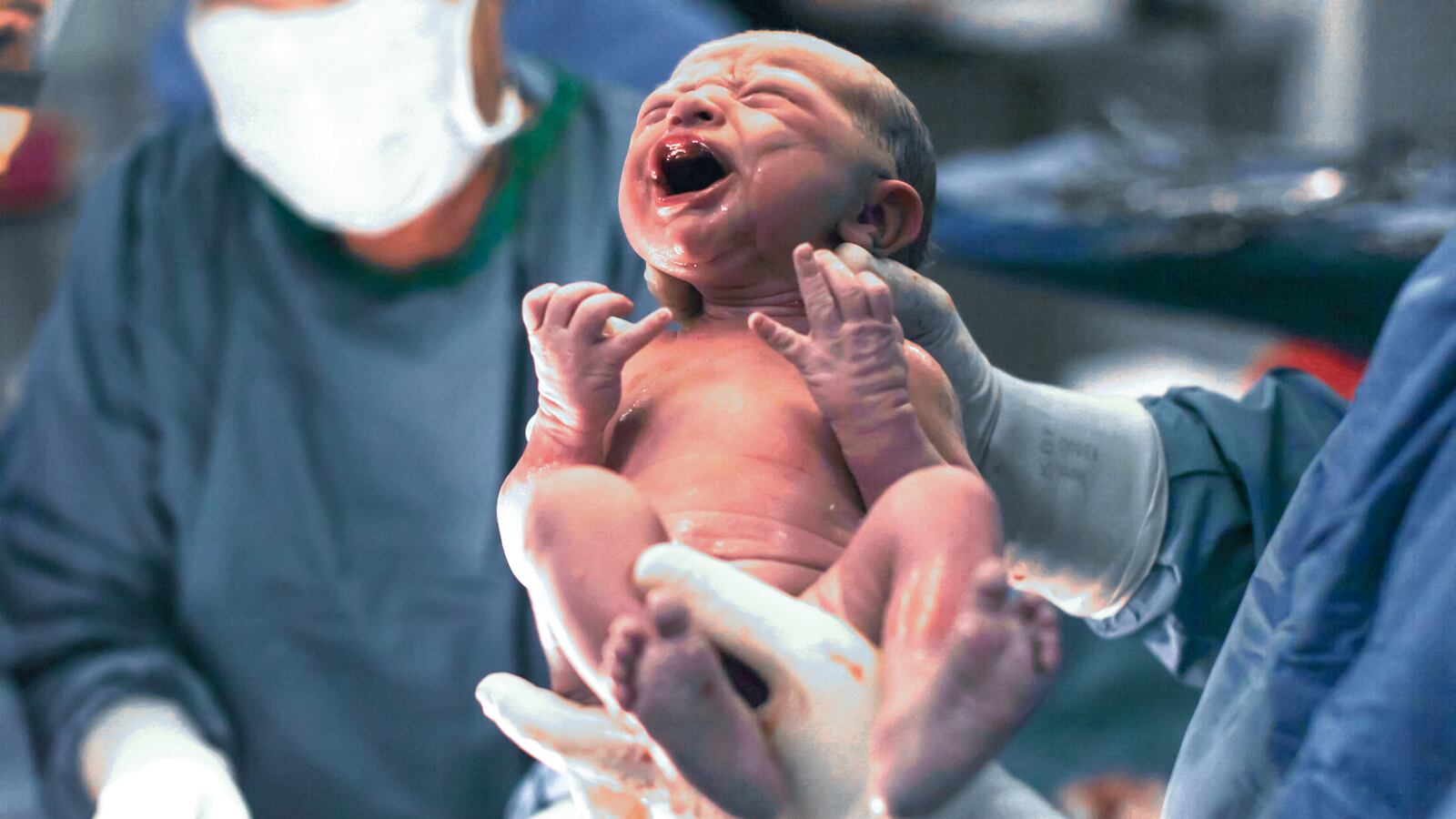
You may not know it, but you likely learned about the importance of Cesarean sections in high school English class. In William Shakespeare’s Macbeth, the eponymous antihero boasts of a prophecy foretelling that he cannot be killed by anyone “woman born.” He’s in for a real surprise when he is ultimately slain by Macduff, who was “from his mother's womb / Untimely ripped”—meaning he was born via C-section.
What was a plot device in the 17th century is now a widely performed, life-saving procedure today, accounting for over 20 percent of childbirths according to the World Health Organization. However, in the U.S., the procedure takes place more often than medically necessary, leading to unnecessary complications and disrupting the collection of microorganisms called the microbiome that live in and on a newborn. A new study published Tuesday in Nature Communications found that these disruptions have lasting impacts, altering the way that children’s immune systems then respond to routine vaccination.
We’ve known for a while that the microbiomes of C-section babies differ from those of newborns born vaginally. Research conducted by Rutgers University microbial ecologist Maria Gloria Dominguez Bello has demonstrated that newborns delivered vaginally acquire some of the same bacterial species found in their mother’s vagina, while C-section babies acquire bacteria present on their mother’s skin. Bello and her co-authors found that swabbing these newborns with gauze that had been put inside the mother’s vagina for an hour led to microbiomes that more closely resembled those of vaginally delivered babies.
Importantly, C-section babies are about 1.2 times likelier than vaginally delivered babies to develop childhood asthma, a complex autoimmune disease. Research has tied this risk to infants’ gut microbiomes: One 2020 study published in Science Translational Medicine found that C-section babies’ asthma risk increased when their gut bacteria did not develop normally a year after birth.
In the most recent study led by researchers in the Netherlands, the authors of the paper followed 120 infants in their first 18 months of life and took saliva samples to gauge their immune responses to the normal Dutch schedule of childhood vaccinations. They found significant differences in the concentration of antibodies present after the children’s meningococcal vaccine, which is recommended at 14 months of age. Those delivered via C-section were found to produce lower levels of IgG antibodies following vaccination than those delivered vaginally—a twofold difference in some instances
To be clear, mode of delivery was not the only factor that played into the development of the immune system. Sex, household pets, antibiotic use, and feeding type (formula versus breastmilk) also impacted the infants’ future response to vaccines.
But the results do suggest that early life gut microbiome development “may set the stage for robust immune responses to childhood vaccinations,” the researchers wrote in the study. In particular, the first 100 days of life constitute a “critical window of opportunity” when the immune system is most impacted by the microbes hanging out in and on a person. Future studies, like those that restore some of the mother’s vaginal microbiome to newborn C-section babies, should follow infants to see if the procedure boosts vaccine response, the authors added.