The experimental drug President Trump received at the White House hours after being diagnosed with COVID-19 was provided under a “compassionate use” program usually reserved for patients who exhaust other treatment options, the manufacturer said Friday.
News of the president consuming the so-called “polyclonal antibody cocktail,” made by New York-based pharma Regeneron, broke around the same time the administration announced Trump was being taken to Walter Reed National Military Medical Center in Maryland for treatment.
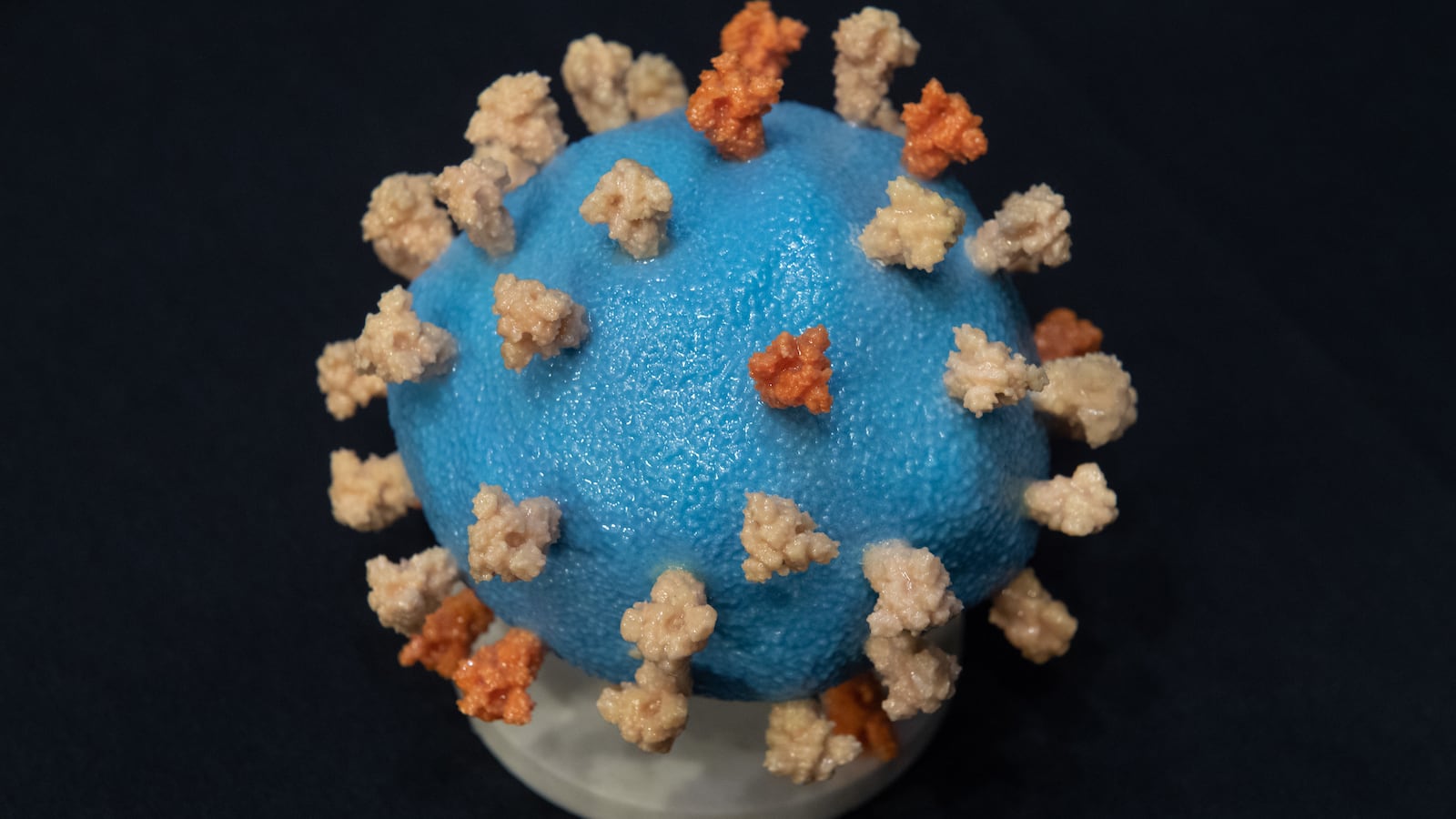
The therapy in question, which some developers refer to as “monoclonal,” combines two lab-grown proteins that attach to the spike proteins on the surface of the novel coronavirus. In theory, this can prevent the pathogen from adhering to the patients’ own cells and causing an infection. It might also help to reduce the severity of symptoms in people who’ve already been infected.
The experimental COVID antibody cocktail itself isn’t terribly controversial, but the circumstances surrounding Trump’s dosing could be.
“This certainly raises questions,” David Ostrov, a professor in the Department of Pathology, Immunology, and Laboratory Medicine at the University of Florida College of Medicine, told The Daily Beast. “Was the president administered an unapproved drug?”
The U.S. Food and Drug Administration’s expanded-access program, also known as “compassionate use,” allows the agency to give unproven experimental treatments to patients whose illnesses are immediately life-threatening. Regeneron told The Daily Beast in a statement that Trump received a compassionate-use exception from the FDA.
But it’s not clear how sick Trump is, and thus whether he should have been eligible for the exception in the first place. “I am not sure the president would qualify for the compassionate-use program, because the patient must have already exhausted available approved treatment options,” Ostrov said. The FDA didn’t immediately respond to a request for comment.
The 74-year-old Trump, who revealed early Friday morning that he and first lady Melania Trump had tested positive for SARS-CoV-2, received a single eight-gram dose of the antibody cocktail, Sean Conley, the White House physician, stated in a Friday memo.
“The president remains fatigued but in good spirits,” Conley wrote.
The Regeneron drug cocktail is still in testing—enrollment of 2,000 test subjects started in August—and experts told The Daily Beast it’s way too soon to know for sure whether it’s safe, or whether it works as advertised.
But these same experts expressed optimism that the antibody cocktail would eventually be useful. The underlying science is sound, they said.
“Targeting the spike with antibodies makes scientific sense, because the spike is what allows the virus to attach to and infect a cell,” argued Otto Yang, an expert in infectious diseases at UCLA.
The same theory is guiding development of several leading novel-coronavirus vaccine candidates. “The underlying mechanisms are similar,” Jeffrey Klausner, a professor of medicine and public health at UCLA who previously worked at the U.S. Centers for Disease Control and Prevention, told The Daily Beast.
“We believe our data will have positive implications for the likelihood of success of vaccines that target the same spike protein that REGN-COV2 targets,” CEO Leonard Schleifer told investors on Tuesday.
U.K. pharma AstraZeneca’s ChAdOx1 nCoV-19 vaccine, one of six that the U.S. government is backing as part of the $13-billion Operation Warp Speed, targets the spike protein, as does the highly experimental vaccine from Novavax in Maryland.
Regulators paused AstraZeneca’s U.S. trials last month after a test subject in the U.K. fell ill with spinal inflammation. It’s unclear whether the non-fatal illness was related to the vaccine. Those U.S. trials have yet to resume.
Likewise, for all its promise, the antibody cocktail Trump has taken is still an unknown quantity.
As Dmitry Korkin, a bioinformatics researcher at the Worcester Polytechnic Institute in Massachusetts put it, the president’s case “must be serious enough for them to resort to a treatment that is only about to enter large-scale clinical trials now.”