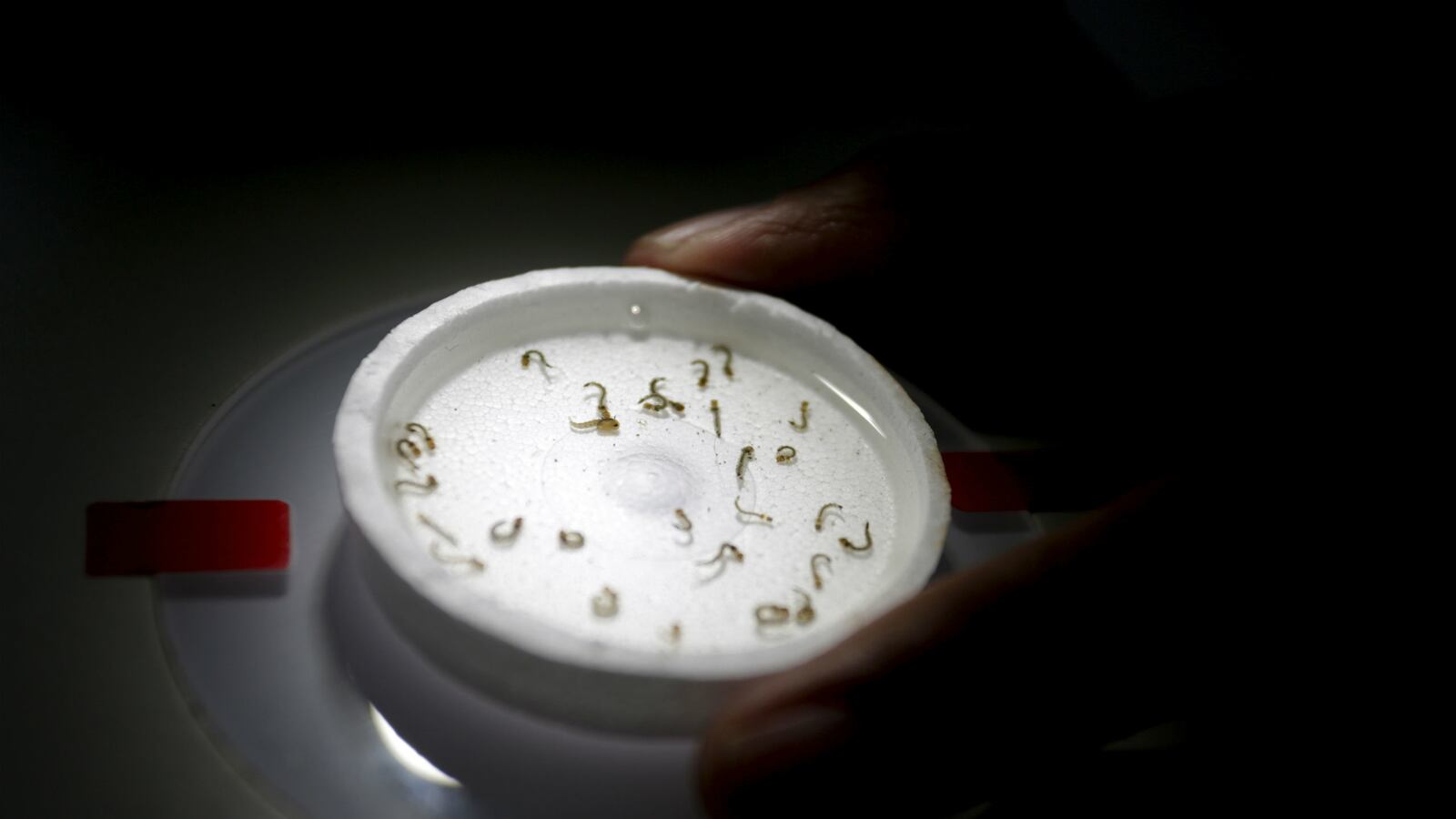
The Food and Drug Administration granted emergency-use authorization Thursday for America’s first commercial test to diagnose the mosquito-borne Zika virus. The test, developed by Quest Diagnostics, uses the same method that government labs use to identify the virus in a patient’s blood. However, the test only works on a person who is currently infected and cannot detect evidence of past infections. The new test costs $500 and can give doctors results in three to five days. Prior to the FDA’s approval, the only Zika tests authorized under the emergency-use authorization were used by CDC-designated labs, Quest officials said. The virus is linked to causing severe birth defects such as microcephaly.
Read it at NBC News