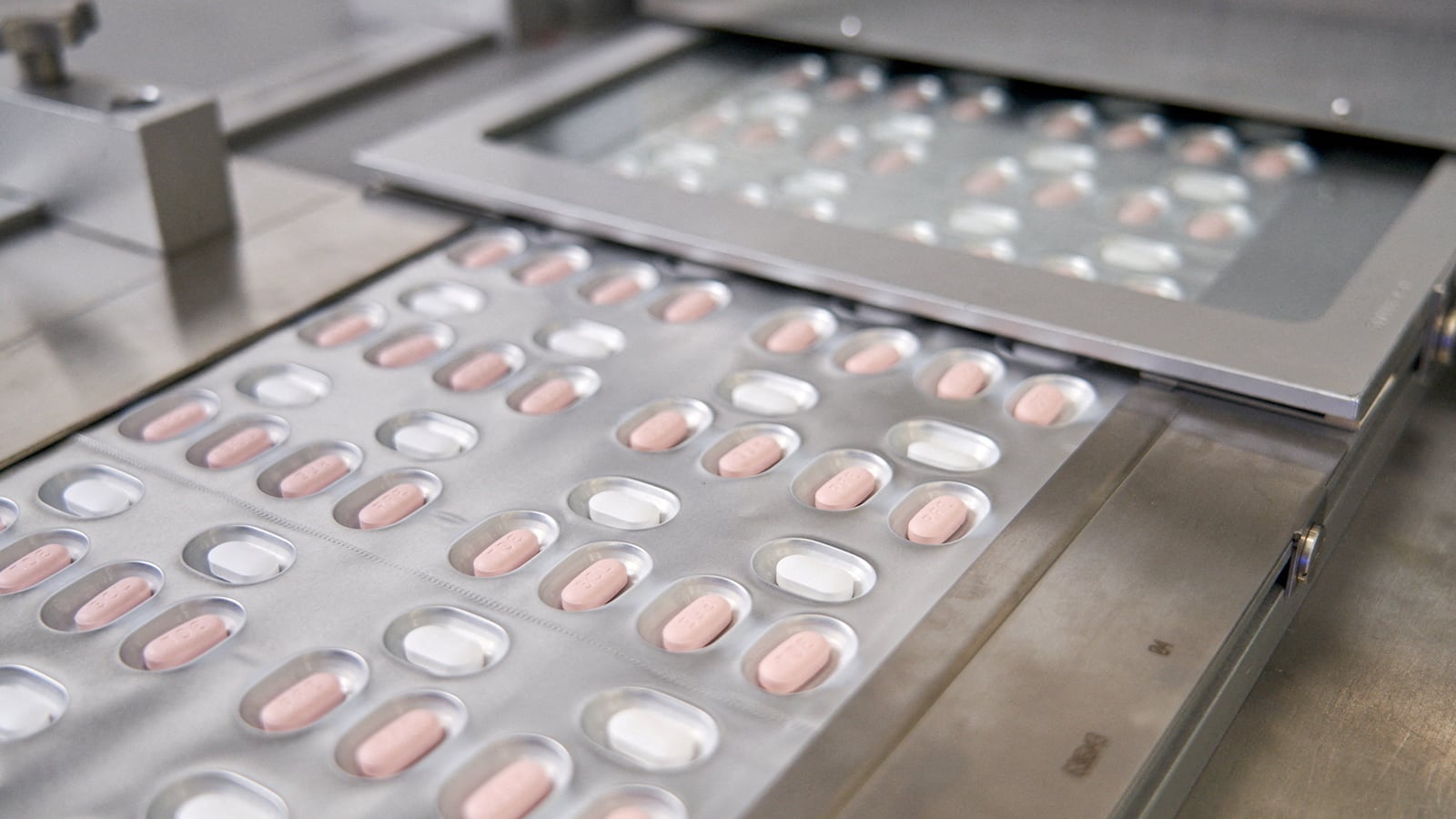
The FDA approved the first at-home treatment option for COVID-19 Wednesday, signifying a major step at fighting the virus even as the Omicron variant surges. The pill, produced by Pfizer, was approved for those aged 12 and up who test positive for the virus and face a higher risk of hospitalization. Trials showed a nearly 90 percent reduction in hospitalizations and deaths for patients most likely to get severe disease. It’s also expected to be effective against Omicron, as it does not target the spike protein that is most heavily mutated by the variant. “The efficacy is high, the side effects are low and it’s oral. It checks all the boxes,” Dr. Gregory Poland of the Mayo Clinic told the Associated Press. “You’re looking at a 90 percent decreased risk of hospitalization and death in a high-risk group—that’s stunning.” The treatment is only effective five days after symptoms first appear, though, troubling some scientists who worry people will not be able to access it within the narrow window.
Read it at Associated Press