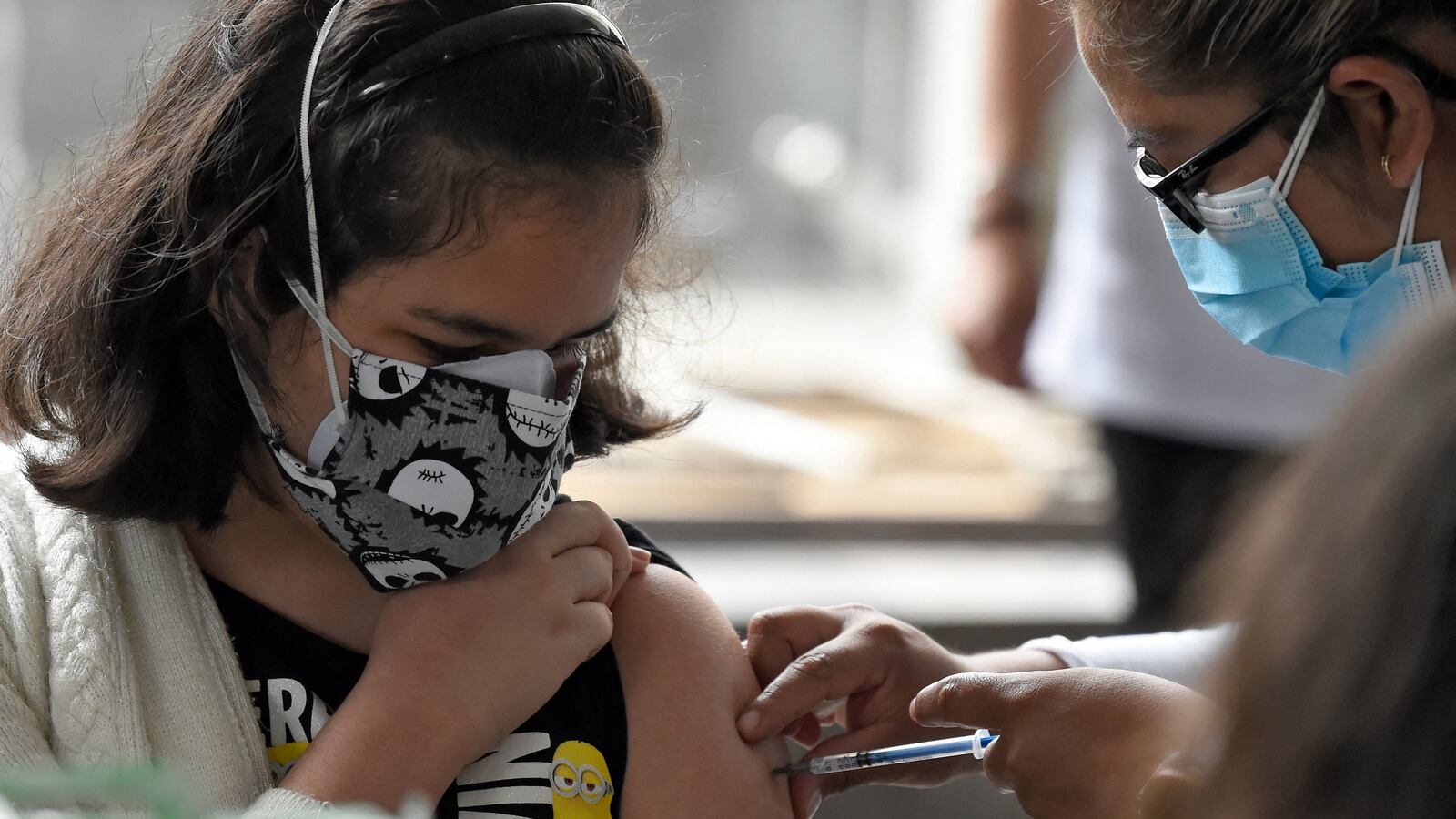
A FDA advisory committee has recommended a lower dose of the Pfizer-BioNTech vaccine against COVID-19 for children aged 5 to 11. The non-binding decision was made after a debate and vote, with 17 members of the panel in favor of the endorsement and one member abstaining. The Tuesday announcement marks the next step in getting jabs to up to 28 million kids ahead of a looming holiday season and a growing number of delta variant cases, according to CNBC.
A report presented to the committee found that the Pfizer vaccine was more than 90 percent effective at protecting kids aged 5 to 11 from coronavirus infection. That cohort makes up about 9 percent of all coronavirus cases in the U.S., the report said. “If I were a parent of a child in this age group, I would want to have the choice,” a member of the advisory board said during the discussion. The recommendation will now be passed to the desk of FDA acting commissioner Janet Woodcock and then to the CDC. The White House said it’s poised to begin distribution as soon as it’s approved.