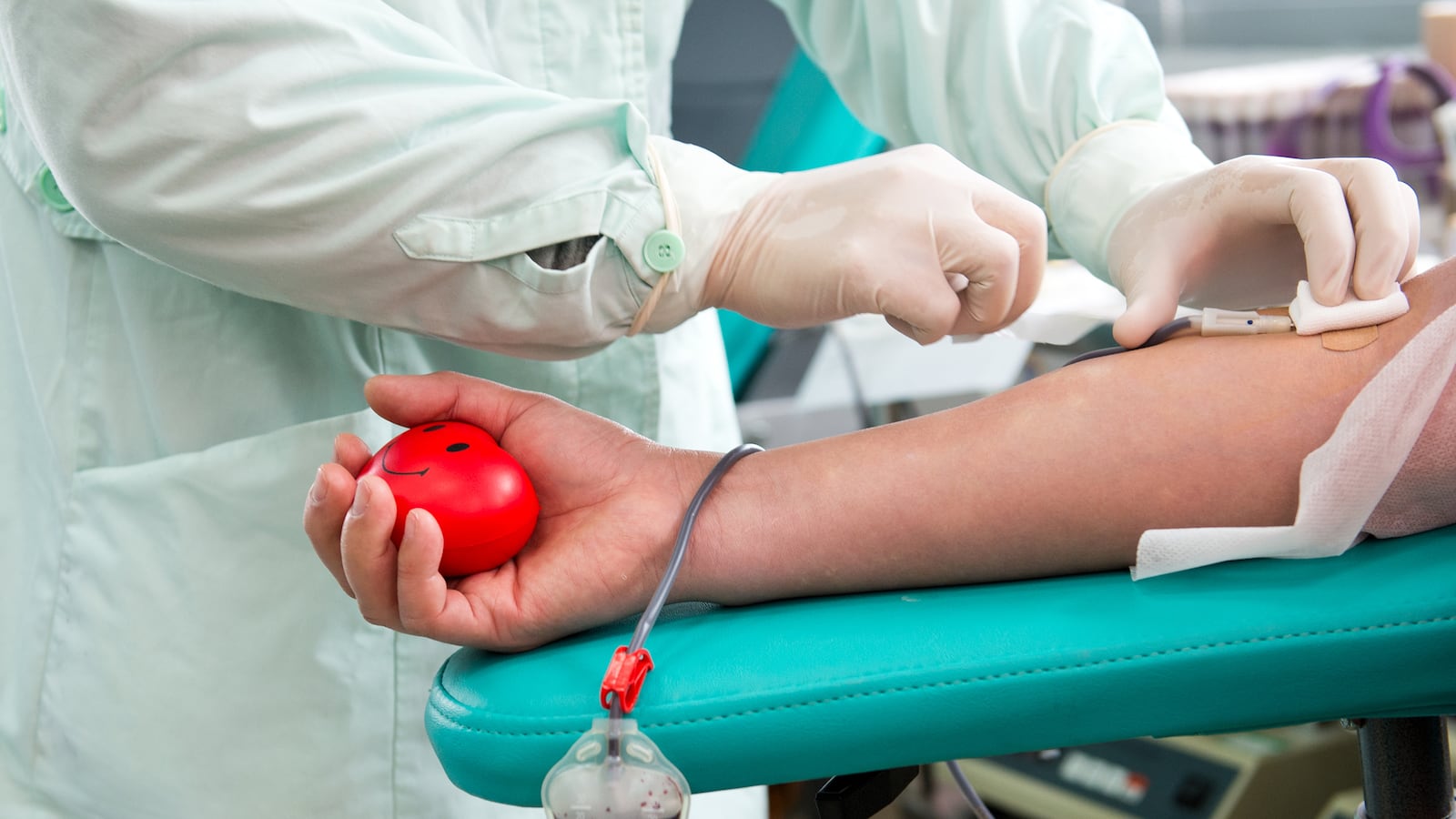
Logan Casey gave blood because he felt it was the right thing to do. A 29-year-old student at the University of Michigan, Casey has been a regular blood donor his entire adult life, donating once every 60 days.
“We all have friends and family members who could benefit from our ability to donate blood,” Casey said.
That changed last October, when a Red Cross staffer asked if he had ever donated under another name. Casey, who is transgender, explained that he had donated under his birth name prior to his transition. After learning Casey was trans, staffers told him that he would be listed as female in his file from that point forward. It was policy, they said.
The center subsequently forced Casey to redo his screening paperwork, asking him to fill in a new form with “distressing” questions about his presumed female anatomy. One question inquired whether he was pregnant.
“The staff member was trying her best but clearly didn't have the language to talk about it,” Casey told The Daily Beast. “That was really frustrating.”
Casey filed a complaint with the Red Cross’s customer service team in November, and the organization quickly replied. He said it was clear, however, that the Red Cross has “no idea what’s going on with trans people.”
“There’s no specific reference to transgender people in our materials,” read the nonprofit’s reply to Casey. “There is with respect to male/male sexual contact. If such has taken place, the individual will be deferred for a 12-month period from the date of last contact. In addition, anyone identifying as a transgender must have undergone the sex change operation. However, during the registration process, all individuals must document as their original birth gender.”
Casey later spoke over the phone with a Red Cross staffer. “[It’s an] FDA-directed policy, and the Red Cross has to follow that,” said the representative, as Casey recalls. The email thus stood.
***
Those policies are reportedly about to change. In December, the FDA updated its official recommendation to stipulate that transgender donors should self-identify their gender on all documentation. Later this year, the Red Cross will release a guidance document that will reportedly state that transgender donors may answer gender-related questions based on the gender with which they identify.
The FDA revisions will be released at a crucial moment for the organization, which is not known for LGBT inclusion.
Until last December, the FDA banned men who have sex with men (MSMs) from donating blood altogether. The FDA announced it would be shortening the deferral period to 12 months after the last instance of same-sex sexual contact. This update was based on improved testing methods for HIV, which have shortened the window for being able to detect the virus.
The policy was widely criticized by LGBT activists, who argued that the blood ban should be lifted entirely, since all donated blood is screened for HIV and other blood-borne pathogens as a matter of policy.
According to recent reports, the FDA is considering doing just that. In July, Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, told CNN that the organization plans to “review [its] donor deferral policies to ensure they reflect the most up-to-date scientific knowledge.”
“Specifically, the FDA invites comments on the feasibility of moving from the existing time-based deferrals related to risk behaviors to alternate deferral options, such as the use of individual risk assessment,” Marks said. “The agency also invites comments regarding the design of potential studies to evaluate the feasibility and effectiveness of such alternative deferral options.”
This could mean finally lifting the 33-year-old ban on MSM donation during a pivotal year for the LGBT community. Following the attack on Orlando’s Pulse nightclub in June, gay and bisexual men who wanted to give blood were often turned away from donation centers.
But as trans activists, writers, and community organizers told The Daily Beast, those changes are long overdue. Throughout the history of the FDA, transgender people report being misgendered, shamed, and stigmatized by blood donation officials because of their gender identity. It’s time, as they argue, to bring the FDA into the 21st century.
When LaSia Wade, founder of the Tennessee Transgender Justice Project, attempted to donate to a local blood donation center in March, she was turned away. The issue was that the gender on her identification didn’t match her physical appearance, which outed her as trans.
“I was distraught,” she told The Daily Beast. “It was disgusting to me that a person could not give blood to help another human. I felt like an alien.”
Andre Perez, the director of America in Transition, a documentary web series about social change from trans perspectives, said his experience was similar. Perez, 28, attempted to donate in 2010, describing the process of giving blood as ridden by misinformation and miscommunication.
“There was this very circular conversation,” Perez said. “Toward the end of it, three people had been brought in.”
When Red Cross staffers discovered he was transgender, Perez says he was repeatedly asked if he had ever had intercourse with another man. He had not. Perez was then asked if he had ever “had sex with a man who had sex with a man.” He had not.
Nonetheless, Perez was still barred from donation. Staffers claimed that they couldn’t “verify [his] HIV risk factors.”
Perez said that the “ignorance” of this line of questioning was “obvious.”
“It doesn’t really get to a lot of questions about HIV—how many sexual partners you’ve had, when the last time you’ve been tested was, or all kinds of relevant behavior,” he explained. “I had known there was a ban in place that negatively impacted gay men, but I hadn’t really thought about the expansive ways in which it could be used to exclude other groups of people.”
Those groups include any population the FDA deems a “high risk” for HIV transmission—intravenous drug users, sex workers, and trans women. Until recently, the latter were classified as MSMs.
Brynn Tannehill, a 41-year-old writer and activist, explained that policies classifying transgender women as “men” were established in 1980, three years before the MSM blood ban was enacted. The Diagnostic and Statistical Manual of Mental Disorders III was released the same year, which was based off research from 1974.
This was an era when trans people were thought of as “perverts, deviants, and mentally ill,” she explained.
“It’s demoralizing to have your own government tell you that you’re a disease-ridden, deluded gay man,” Tannehill said. “If you said that to somebody’s face, you’re lucky if you get a drink thrown at you.”
While Tannehill argued that these policies assumed that “all trans people are all promiscuous,” Perez explained that the AIDS panic of the Reagan era compounded that stigma. As HIV initially spread throughout the United States, medical professionals knew little about how the disease was transmitted.
A lot has improved since.
“We can now identify HIV-positive blood with a fourth-generation HIV test within a window of a week [of infection],” Perez said. “[Current FDA] policies are not based on actual information about how HIV is transmitted. In a climate where we have scientific clarity around disease transmission, I don’t understand why we’re still relying on stereotypes.”
“Why doesn’t science inform policy?” Perez asked. “Why is policy informed by this fear that we have?”
Perez believes that instead of relying on outdated stigma, the FDA needs to look at individual risk factors in transmission. These factors include the donor’s number of sexual partners and when the individual was last tested for HIV.
***
Although the updated FDA guidelines on trans donors were announced this year, the news received surprisingly little coverage in the mainstream press. Tannehill, who has worked on the blood ban issue for years, was unaware of the modification—and claimed that many other trans people aren’t aware of it either.
“The fact that no one knows about this is not a good thing,” she said.
When the new policies are rolled out in late 2016, Tannehill predicts that blood donation centers will be slow to implement them.
“Because trans women have been treated as MSMs for so long, local centers are still going to be under that impression,” Tannehill argued. “Even if they do find out about [these guidelines], an overabundance of caution is going to allow them to continue to ban transgender people from donating blood.”
Elizabeth Marie Rivera told The Daily Beast that the FDA must consult the trans community to ensure these guidelines are implemented properly.
“They need to be working directly with the trans community and bringing trans-identified individuals into consultation meetings,” said Rivera, a 38-year-old Latina trans activist. “In order to do it correctly, you need to be speaking to us.”
Implementation has already proved to be a problem for the FDA. Following the Orlando shooting, many blood donation centers reported that their systems had not yet been updated to reflect the 12-month window for MSM donors, though the decision had come down six months earlier.
“We have an extremely long way to go,” Rivera said.
In bringing government policy up to date with current medical understanding about queer and transgender lives, Casey said the potential benefit is huge. In 2014, UCLA’s Williams Institute estimated that allowing MSMs to donate would save 1.8 million people. Making it easy, safe, and comfortable for trans people to also give blood would only increase that number.
During the meantime, many blood donation centers across the U.S. are ailing—unable to get the donations they need.
“I’ve been getting an email from the local Red Cross two to three times a week saying that there’s a shortage of blood in our area and they need donors now,” Casey said. “In the big picture, lives are being lost.”