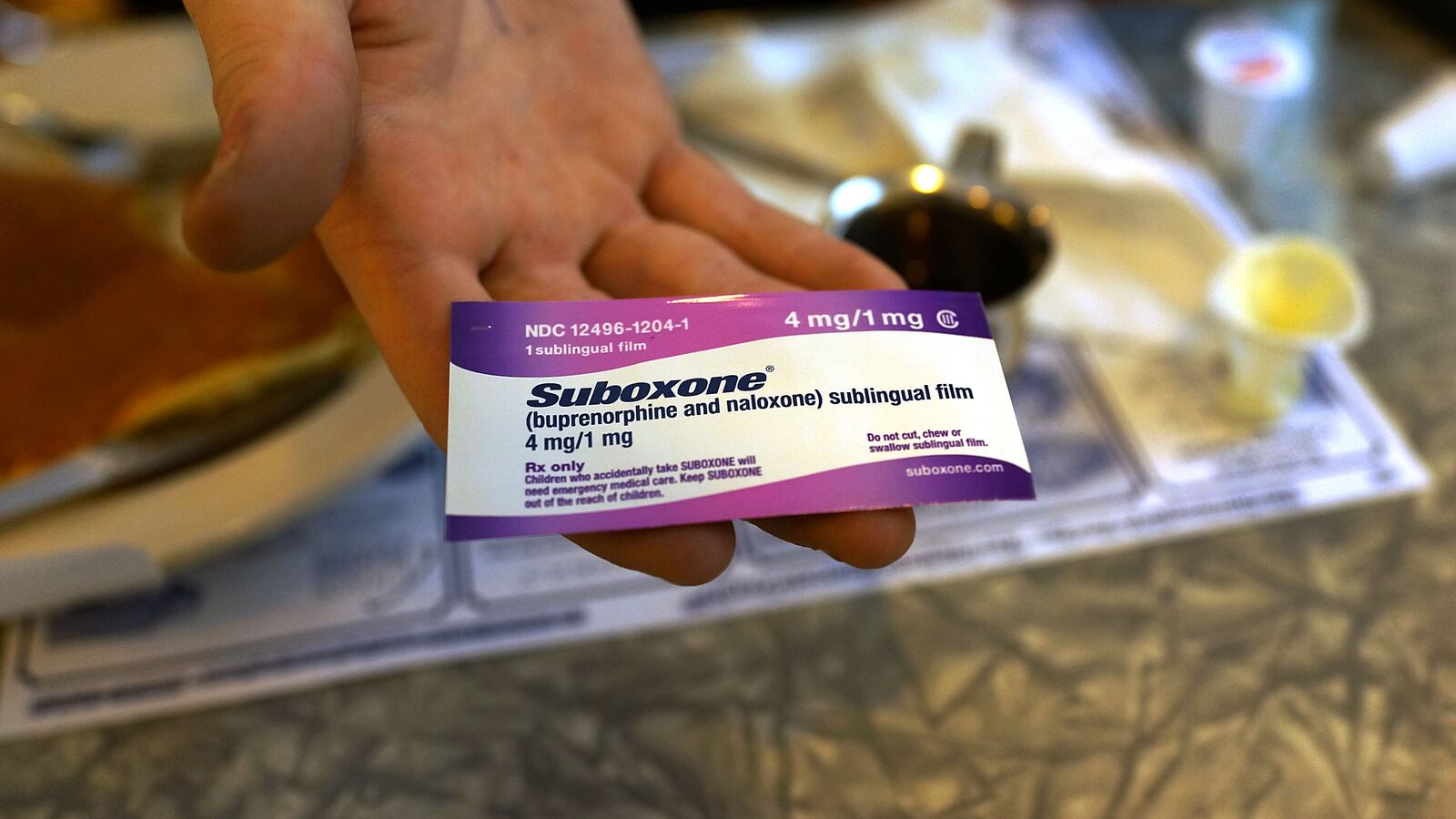
The Justice Department charged pharmaceutical company Indivior with making false statements about its opioid addiction treatment drug and leading opioid addicts to doctors who were prescribing their potent drug under “suspect circumstances.” According to the indictment and a DOJ press release, the company engaged in a scheme to increase the sales of Suboxone Film by marketing the drug as “safer” and “less abusable” than other opioid addiction treatment drugs without any evidence to back up those claims. The company also allegedly established a hotline for opioid addicts that led them to doctors who Indivior knew were prescribing high doses of Suboxone Film to more people than legally permitted and in a “careless and clinically unwarranted manner.” DOJ alleges that Indivior’s practices caused “thousands” of opioid abusers to become addicted to Suboxone Film, incurring a “substantial cost to the government,” as Suboxone Film was covered by state Medicaid programs. The indictment states the company has been charged with “one count of health care fraud, four counts of mail fraud, and twenty-two counts of wire fraud.”
The company released a statement and a list of rebuttals of the DOJ’s allegations on Monday night, saying the indictment was an effort to “pursue self-serving headlines” and that it would “vigorously” contest the case in court. “We are extremely disappointed in this action by the Justice Department, which is wholly unsupported by either the facts or the law,” the company said.