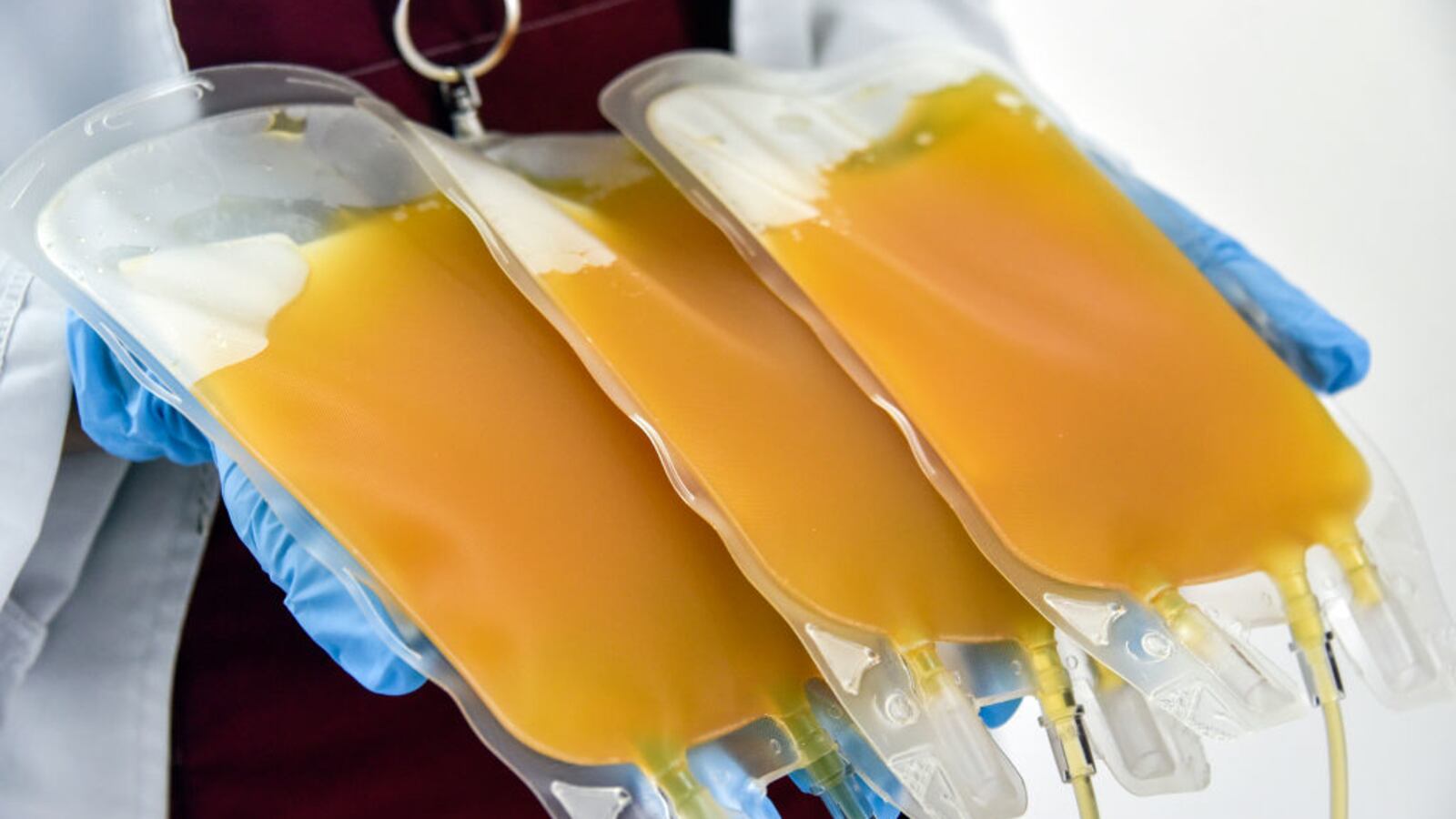
A panel of experts convened by the National Institute of Health issued a statement Tuesday directly contradicting the Food and Drug Administration and the president’s claim that convalescent plasma has been proven to be effective in treating COVID-19 patients. According to the NIH panel, data indicating convalescent plasma can treat COVID-19 is “insufficient.” The FDA granted an emergency authorization for the use of the substance, gleaned from the blood of coronavirus survivors, just last week. President Donald Trump touted a statistic saying that the plasma was “proven to reduce mortality by 35 percent.” Scientists have called that figure misleading because the study that made the finding has not yet been peer reviewed or published, nor did it make use of a control group given a placebo, making it neither randomized nor controlled, the gold standard of medical research. The FDA’s commissioner Stephen Hahn has admitted he overstated convalescent plasma’s benefits as well. The NIH panel found that there were “currently no data from well-controlled, adequately powered randomized clinical trials” and therefore “insufficient data to recommend either for or against the use of convalescent plasma for the treatment of COVID-19.”
Read it at BuzzFeed News