By Hemant Khanna, The Conversation
In recent months, even as our attention has been focused on the coronavirus outbreak, there have been a slew of scientific breakthroughs in treating diseases that cause blindness.
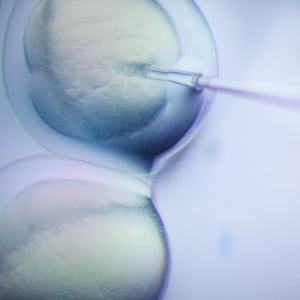
Researchers at U.S.-based Editas Medicine and Ireland-based Allergan have administered CRISPR for the first time to a person with a genetic disease. This landmark treatment uses the CRISPR approach to a specific mutation in a gene linked to childhood blindness. The mutation affects the functioning of the light-sensing compartment of the eye, called the retina, and leads to loss of the light-sensing cells.
According to the World Health Organization, at least 2.2 billion people in the world have some form of visual impairment. In the United States, approximately 200,000 people suffer from inherited forms of retinal disease for which there is no cure. But things have started to change for good. We can now see light at the end of the tunnel.
I am an ophthalmology and visual sciences researcher, and am particularly interested in these advances because my laboratory is focusing on designing new and improved gene therapy approaches to treat inherited forms of blindness.
Gene therapy involves inserting the correct copy of a gene into cells that have a mistake in the genetic sequence of that gene, recovering the normal function of the protein in the cell. The eye is an ideal organ for testing new therapeutic approaches, including CRISPR. That is because the eye is the most exposed part of our brain and thus is easily accessible.
The second reason is that retinal tissue in the eye is shielded from the body’s defense mechanism, which would otherwise consider the injected material used in gene therapy as foreign and mount a defensive attack response. Such a response would destroy the benefits associated with the treatment.
In recent years, breakthrough gene therapy studies paved the way to the first ever Food and Drug Administration-approved gene therapy drug, Luxturna TM, for a devastating childhood blindness disease, Leber congenital amaurosis Type 2.
This form of Leber congenital amaurosis is caused by mutations in a gene that codes for a protein called RPE65. The protein participates in chemical reactions that are needed to detect light. The mutations lessen or eliminate the function of RPE65, which leads to our inability to detect light—blindness.
The treatment method developed simultaneously by groups at University of Pennsylvania and at University College London and Moorefields Eye Hospital involved inserting a healthy copy of the mutated gene directly into the space between the retina and the retinal pigmented epithelium, the tissue located behind the retina where the chemical reactions takes place. This gene helped the retinal pigmented epithelium cell produce the missing protein that is dysfunctional in patients.
Although the treated eyes showed vision improvement, as measured by the patient’s ability to navigate an obstacle course at differing light levels, it is not a permanent fix. This is due to the lack of technologies that can fix the mutated genetic code in the DNA of the cells of the patient.
Lately, scientists have been developing a powerful new tool that is shifting biology and genetic engineering into the next phase. This breakthrough gene editing technology, which is called CRISPR, enables researchers to directly edit the genetic code of cells in the eye and correct the mutation causing the disease.
Children suffering from the disease Leber congenital amaurosis Type 10 endure progressive vision loss beginning as early as one year old. This specific form of Leber congenital amaurosis is caused by a change to the DNA that affects the ability of the gene—called CEP290—to make the complete protein. The loss of the CEP290 protein affects the survival and function of our light-sensing cells, called photoreceptors.
One treatment strategy is to deliver the full form of the CEP290 gene using a virus as the delivery vehicle. But the CEP290 gene is too big to be cargo for viruses. So another approach was needed. One strategy was to fix the mutation by using CRISPR.
The scientists at Editas Medicine first showed safety and proof of the concept of the CRISPR strategy in cells extracted from patient skin biopsy and in nonhuman primate animals.
These studies led to the formulation of the first ever in human CRISPR gene therapeutic clinical trial. This Phase 1 and Phase 2 trial will eventually assess the safety and efficacy of the CRISPR therapy in 18 Leber congenital amaurosis Type 10 patients. The patients receive a dose of the therapy while under anesthesia when the retina surgeon uses a scope, needle and syringe to inject the CRISPR enzyme and nucleic acids into the back of the eye near the photoreceptors.
To make sure that the experiment is working and safe for the patients, the clinical trial has recruited people with late-stage disease and no hope of recovering their vision. The doctors are also injecting the CRISPR editing tools into only one eye.
An ongoing project in my laboratory focuses on designing a gene therapy approach for the same gene CEP290. Contrary to the CRISPR approach, which can target only a specific mutation at one time, my team is developing an approach that would work for all CEP290 mutations in Leber congenital amaurosis Type 10.
This approach involves using shorter yet functional forms of the CEP290 protein that can be delivered to the photoreceptors using the viruses approved for clinical use.
Gene therapy that involves CRISPR promises a permanent fix and a significantly reduced recovery period. A downside of the CRISPR approach is the possibility of an off-target effect in which another region of the cell’s DNA is edited, which could cause undesirable side effects, such as cancer. However, new and improved strategies have made such likelihood very low.
Although the CRISPR study is for a specific mutation in CEP290, I believe the use of CRISPR technology in the body to be exciting and a giant leap. I know this treatment is in an early phase, but it shows clear promise. In my mind, as well as the minds of many other scientists, CRISPR-mediated therapeutic innovation absolutely holds immense promise.

In another study just reported in the journal Science, German and Swiss scientists have developed a revolutionary technology, which enables mice and human retinas to detect infrared radiation. This ability could be useful for patients suffering from loss of photoreceptors and sight.
The researchers demonstrated this approach, inspired by the ability of snakes and bats to see heat, by endowing mice and postmortem human retinas with a protein that becomes active in response to heat. Infrared light is light emitted by warm objects that is beyond the visible spectrum.
The heat warms a specially engineered gold particle that the researchers introduced into the retina. This particle binds to the protein and helps it convert the heat signal into electrical signals that are then sent to the brain.
In the future, more research is needed to tweak the ability of the infrared sensitive proteins to different wave lengths of light that will also enhance the remaining vision.
This approach is still being tested in animals and in retinal tissue in the lab. But all approaches suggest that it might be possible to either restore, enhance or provide patients with forms of vision used by other species.
Hemant Khanna is an associate professor of ophthalmology at the University of Massachusetts Medical School