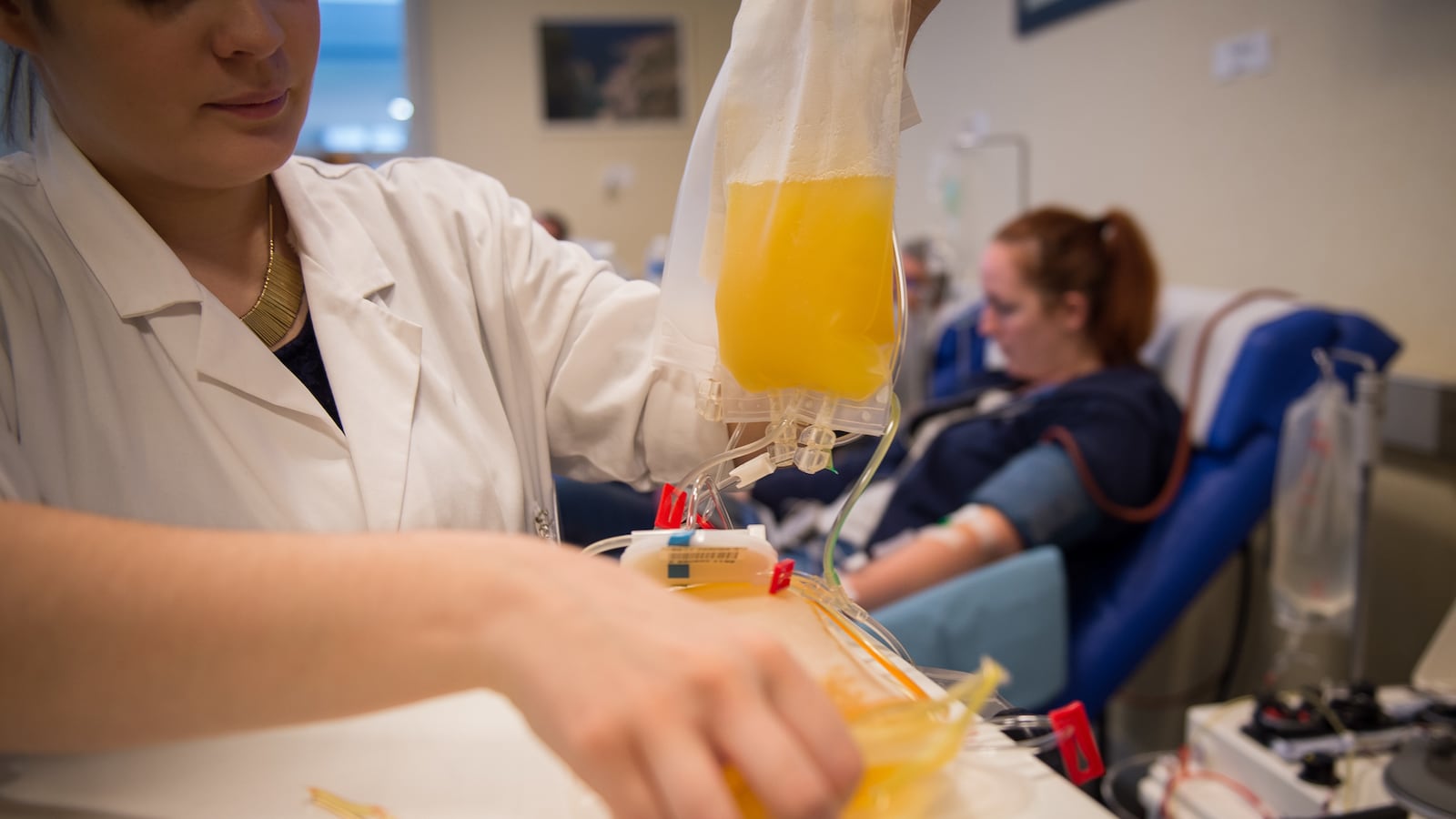
The World Health Organization on Monday was cautious against endorsing the expanded use of antibody-rich blood plasma to help COVID-19 patients, stating that evidence it works as a treatment remains “low quality.” The statement comes just one day after the Food and Drug Administration on Sunday gave emergency approval for expanded use of convalescent plasma after President Donald Trump blamed the agency for blocking the release of vaccines and therapeutics for political reasons.
“At the moment, it’s still very low-quality evidence,” WHO chief scientist Soumya Swaminathan said in a news conference, noting only a few clinical trials have produced results and the evidence so far is not convincing enough to endorse. “So we recommend that convalescent plasma is still an experimental therapy, it should continue to be evaluated in well-designed randomized clinical trials.”
Swaminathan added that while some trials have shown benefits, their small test sizes yielded inconclusive results. Another challenge is plasma’s variability—since it’s drawn from various individuals. World Health Organization senior adviser Bruce Aylward also stressed that there are potential safety risks because “there are a number of side effects” ranging from mild fevers to severe lung injuries.